Discovery of peptide inhibitors targeting hPD-1
Activation of the immune system for cancer therapy has long been a goal in immunology and oncology,
and a recent breakthrough in cancer immunotherapy is the immunologic checkpoint blockade . One of the
checkpoint receptors is hPD-1. Using the de novo peptide design method, we designed some hPD-1 binding peptides with five identified residues Y56, R113, A121, D122 and Y123 derived from cognate ligand hPD-L1 as anchors. (figure1). The most potent peptide Ar5Y_4 showed a KD value of 1.38 ± 0.39 μM, comparable to the binding affinity of the cognate hPD-L1. A Surface Plasmon Resonance (SPR) competitive binding assay result indicated that Ar5Y_4 could inhibit the interaction of hPD-1/hPD-L1 (figure2). Moreover, peptide Ar5Y_4 can restore the suppressed function of Jurkat T cells by blocking the
interaction of hPD-1 and hPD-L1. (figure3). So peptide Ar5Y_4 is a promising inhibitor and can be utilized as the starting point for further leads optimization..
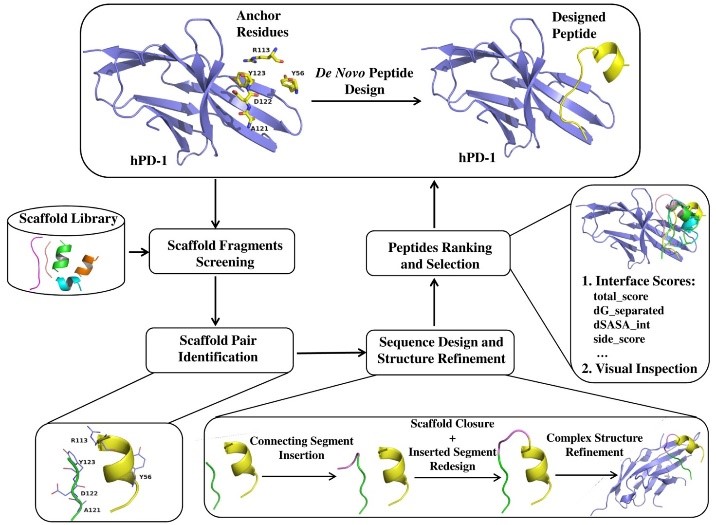
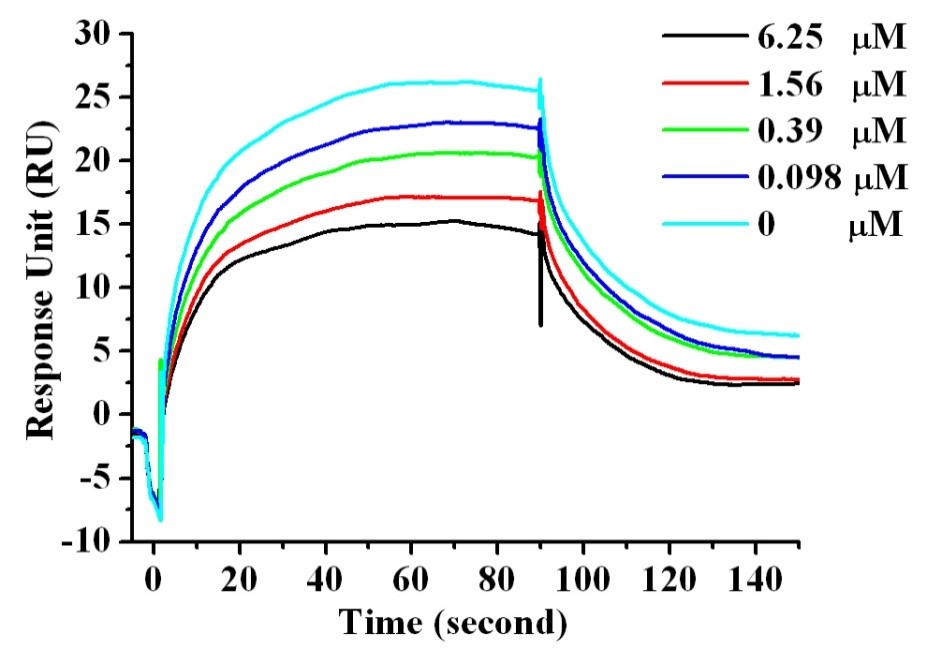
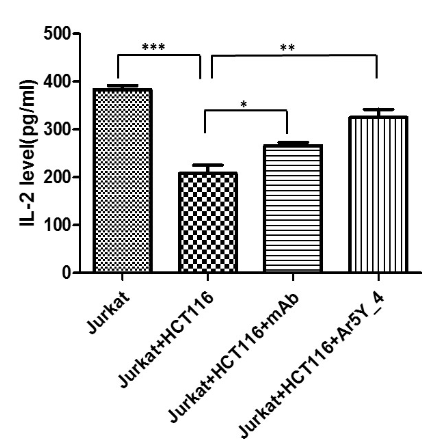
Figure1 Figure2 Figure3
User Login
Web Services
Contact us
Honglin Li's Lab
Shanghai Key Laboratory of New Drug Design
School of Pharmacy
East China University of Sci. & Tech.
Room 527, Building 18, 130 Meilong Road,
Shanghai, 200237, P. R. China
Tel: (86) 21 6425 0213 Prof. Honglin Li
hlli@ecust.edu.cn
Copyright © 2026 Prof. HongLin Li's Group, School of Pharmacy, East China University of Science & Technology · All Right Reserved.
沪ICP备19004698号-1 |  沪公网安备31011302004713号
沪公网安备31011302004713号